If you set up your initial screens with 100 nl (protein) + 100 nl (reservoir
solution) drops, it's quite likely that you will have to scale up to larger
drops in order to get crystals that are large enough to collect data from.
If you take the same solutions and simply increase the volume to say 1 + 1 ul,
you will often get precipitation instead of crystallization. This is a
counterintuitive result because people correctly believe that the smaller
drops will dry out more quickly, and they therefore assume that they will see
more precipitation in the smaller drops.
However, the situation becomes clear when you realize that there are two
important differences between large and small drops:
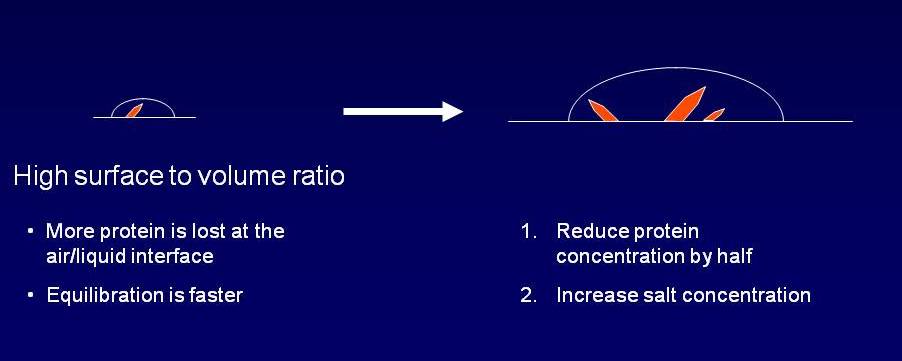
1. The surface area to volume ratio is greater for the smaller drops (to put it
another way, more of the small drop is close to the surface).
2. The smaller drops equilibrate with the reservoir more quickly.
Point (1) means that more protein is lost on surfaces of
smaller drops. In fact, for a typical protein, using 0.1 + 0.1
drops, roughly half of the protein is denatured and either sticks to the plastic
surface of the plate or to the air-drop interface, forming a skin. You
therefore need to decrease the amount of protein when you scale up.
This can be done either by diluting the protein by a factor of
around
two, or by halving the volume of protein added to the drop, e.g. using
0.75 (protein) + 1.5 (reservoir solution).
An alternative approach is to screen with 0.2 + 0.1
drops in the first place. According to Heather Ringrose (Pfizer, UK), you
find conditions that are different to those with the normal 0.1 + 0.1 dops, but
these new conditions will scale up to 1 + 1.
Many people use 0.3 + 0.3
for their initial screens which is a compromise between several factors:
the need to save protein, the ease of scaling up, and the likelihood of
obtaining diffraction-quality crystals right out of the initial screen. It
should not be necessary to increase the drop volume beyond this for initial
screening.
Incidentally, if you're trying to grow really large crystals (for example for
neutron diffraction) it often helps to increase the volume of drops to 5 + 5 or
above, especially in a microbatch-under-oil setup. This implies that
crystals often stop growing because they've run out of protein.
Another tip is to increase the salt concentration in the scaled-up drop.
If you have e.g. 200 mM salt in the nano-drops, try increasing it to around 300.
Similarly, if the main precipitant is 1 M salt solution, try 1.5 M. This
observation comes from both practical experience and data mining. It's not
clear what the mechanism is, but it may be related to the slower equilibration
rate for large drops.
